The $2.3 Billion Paradox: A Strategic White Paper for Executive Leadership and Venture Capitalists - II
February 12, 2026
Episode II:
The Data of Reality — Separating Signal from Noise
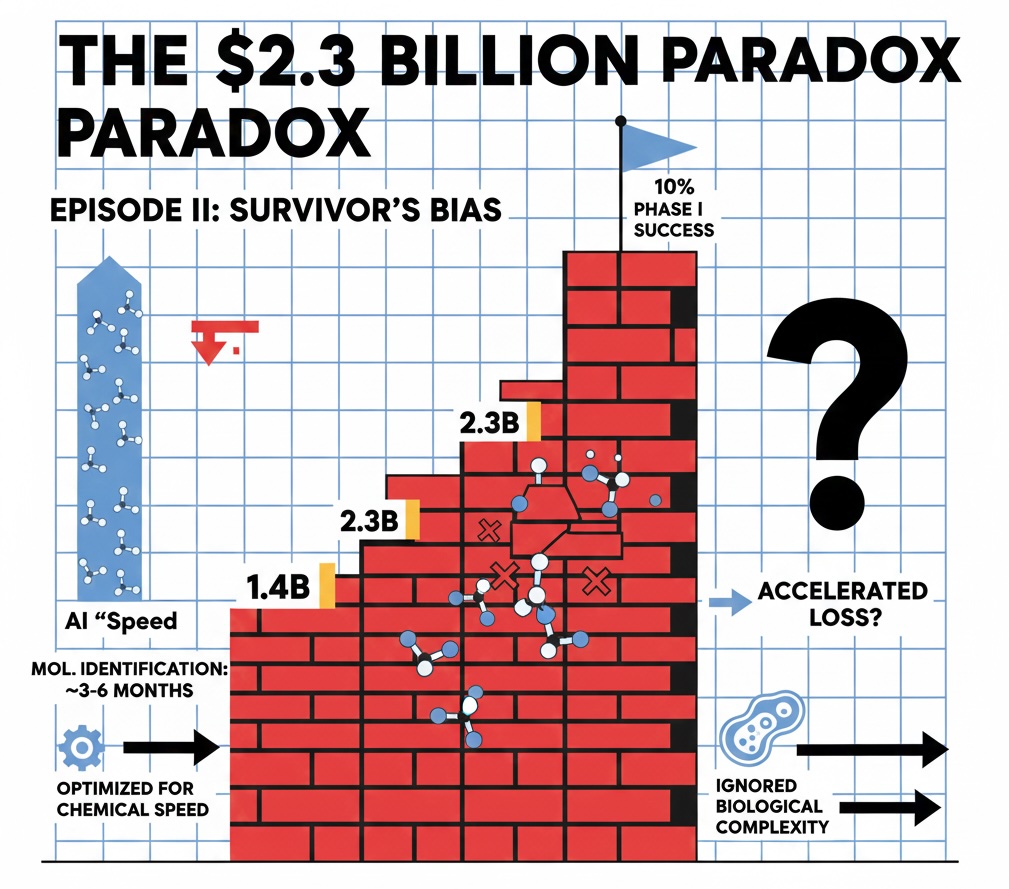
Episode 1 has identified the $2.3 billion efficiency trap as the “Elephant in the Room.” But there is a second, stealthier elephant we need to talk about: Survivorship Bias. It’s easy to get excited about the “poster children” of AI, but observant leaders will notice the graveyard of failures that never make the press release.
The “Convenience Gap”: Where Does the Data Actually Come From?
You’ve likely heard the claim: “AI makes drug discovery 2.5 years faster”. But we have to wonder: Is any of this actually working in humans? Is the above number valid across the board?” To address the first question, let’s compare the status of the top AI-validated assets shown in Table 1, from which you may agree that AI is a surgical tool, rather than a magic wand.
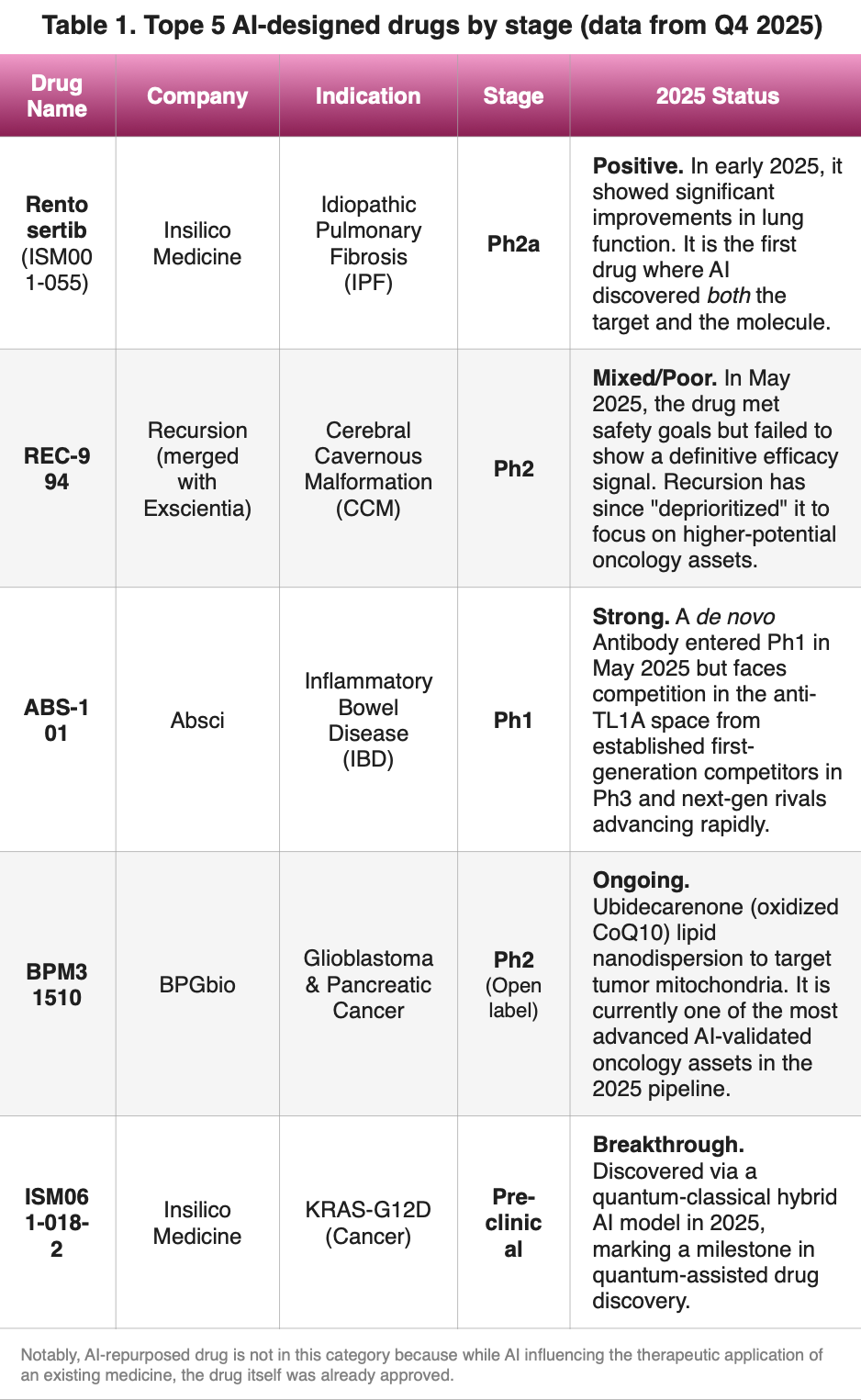
Looking at the table above, the signal becomes clear: AI can predict how a drug binds to a protein, but it still struggles to predict how a human body reacts to that drug.
- The Successes (Insilico): In 2025, Insilico Medicine demonstrated that finding a “novel target” for Fibrosis took 18 months (vs. 4–6 years traditionally). This is the source of the “2.5-year” figure—it’s front-loaded.
- The Reality Checks (Recursion): Remind us that “Discovery” is easy for AI, but “Efficacy” (making the patient better) still requires the gauntlet of human trials.
- The Breakthroughs (Insilico & Absci): Quantum computing and “de novo” design are the frontiers, although in th case of Absci anti-TL1A is already a crowded space.
These data also reveals a hard truth to answer the second question: AI’s prowess is not universal. Models are “data-hungry,” and some areas such as Oncology and Small Molecules are much “friendlier” to AI than others (such as Neuroscience and Biologics). By distinguishing the “Signal” from the “Noise,” it’s not hard to realize that the majority of the venture capital world is currently blinded by Survivorship Bias: We celebrate a few “poster children” while ignoring that most of the AI-biotech firms struggling with the same 70% Phase 2 failure rates as traditional pharma according to data reported by Nature Reviews Drug Discovery (2025).
The Survivorship Bias: Why the 2.5-Year Promise is Risky
One of the most common paradoxes in pharmaceutical economics is that money saved in “Discovery” doesn’t automatically make the massive, traditional clinical trial process any cheaper. If you are a venture capitalist or a government agency, you shouldn’t be looking for “AI companies.” You should be looking for companies that use AI to reduce the risk of Phase 2 failure. Looking at the broader landscape in 2025, there is significant evidence that AI does not yet provide the 2.5-year promise across the board (Table 2):
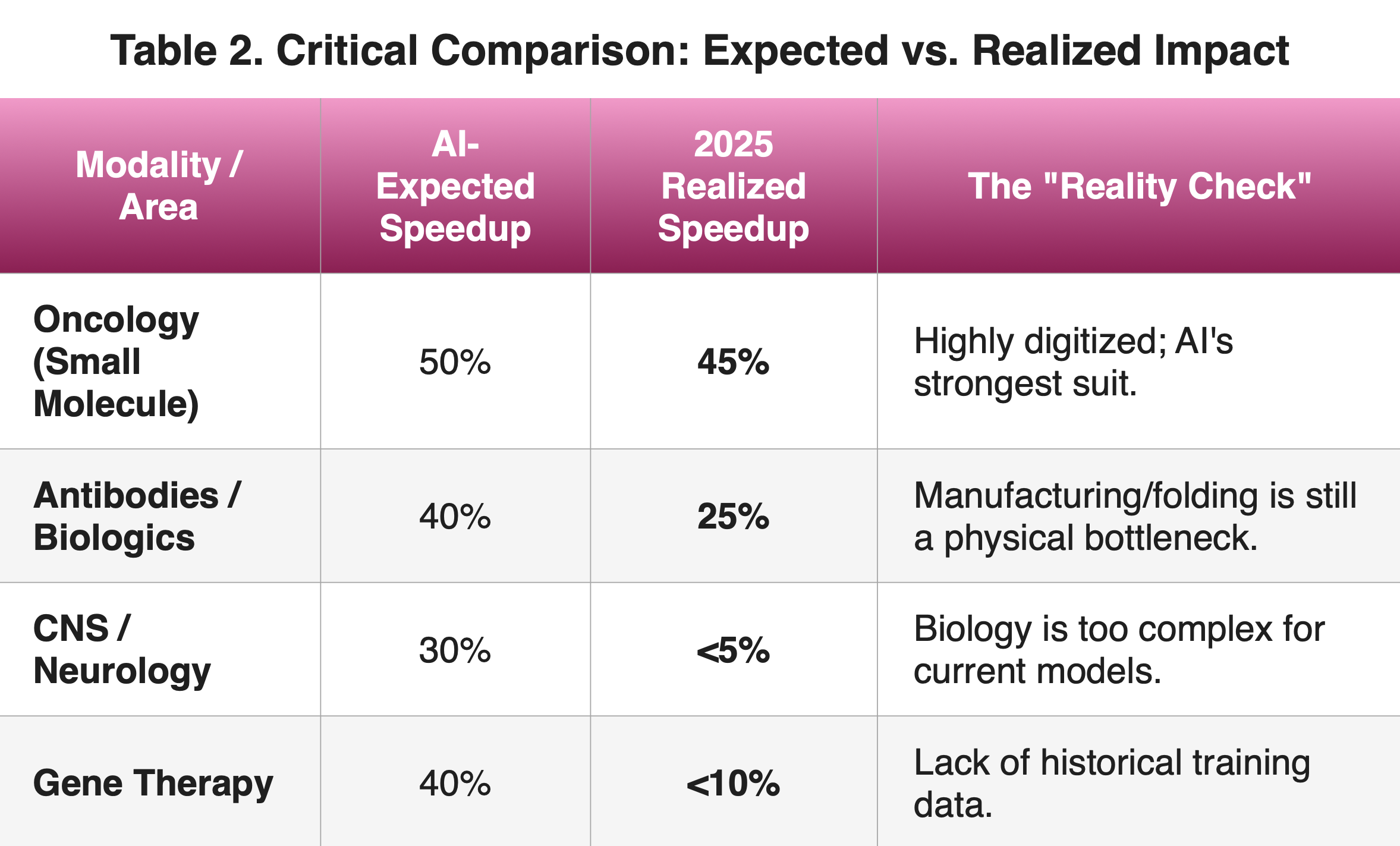
To summarize, this analysis reveals a “bifurcation” in how AI-designed drugs are performing compared to their traditional counterparts; the 2.5-year figure is a best-case scenario for small molecule in one therapeutic area. For the rest of the industry, AI currently acts more like an “advanced calculator”—it makes individual tasks faster, but it hasn’t yet shortened the overall 10-year journey for complex drugs.
To find the “Alpha,” we must look at the tangible evidence.
In our final episode:
We are going to move from “What is happening” to “How you win.” We’ll lay out the roadmap to the $1.4 billion drug—the Holy Grail of R&D—and give you the specific questions you should be asking every biotech CEO who walks into your office.
Disclaimer:
The information provided in this article is for general informational and educational purposes only. It is not intended as, and should not be construed as, professional business, legal, investment, or medical advice. Before making any strategic decisions, you should consult with a qualified professional. Calxera makes no representation or warranty of any kind, express or implied, regarding the accuracy, adequacy, validity, reliability, availability, or completeness of any information on this site.